Research output per year
Research output per year
Flower senescence

Figure 1. Cut flower sales per capita are high in the UK and the range of species available impressive.
Senescence of flowers, as it is for leaves, is the terminal phase of development and limits both the attractiveness of flowers for pollinating insects, thereby limiting seed set, and the vase life of cut ornamentals. The significance of flower senescence is therefore considerable and understanding and manipulating the process is an important goal for plant scientists.
In the UK the value of the cut flower trade is very large (Fig. 1) and approaches that of the music industry and yet cut flowers rarely grab the headlines in the same way. In 2003 sales of cut flowers in the UK averaged £26.00 per person, this is nearly twice that spent by each US citizen but less than spent by Dutch and Swiss residents. Our group is currently investigating the senescence of ornamentals and in the past we have published work on such species as lupin, petunia, carnation, tobacco, iris and daylily. More recently, however, we have focused on Alstroemeria.
Alstroemeria feature as one of the top 10 species of cut flowers but unlike many of the other crops, Alstroemeria can be grown successfully in the UK. However, unlike carnations and many other species, Alstroemeria is not sensitive to ethylene and hence there are no commercially available treatments available to extend their vase-life. The formulations that one finds for lilies and Alstroemeria (Fig. 2) are in fact designed to reduce leaf yellowing which can sometimes be so unsightly that consumers would discard their flowers before the flowers die (Fig. 2). By investigating changes in gene expression during flower senescence in Alstroemeria we have gained valuable knowledge that can be applied to the formulation of new floral preservatives, help design breeding programmes to produce better quality crops, and even provide diagnostic tests to ensure that flowers reaching the market are of an optimum quality.

Figure 2. Special formulations of flower food are available for lilies and Alstroemeria (left) but these are formulated to delay leaf yellowing which can occur in untreated flowers making stems unattractive even before the flowers die (right).
Gene expression controls much of the phenotypic appearance and physiology of flowers (some control being at the post-translational level) and the application of microarrays has enabled us to investigate gene changes without any preconceptions as to which genes might be critical. We have constructed a microarray comprising 6,500 floral genes, some produced by subtractive hybridisation, others by sampling cDNA libraries made from flowers at various stages of development or treated in ways typical of the supply chain. From this array data, a number of genes specific to flower senescence have been identified and others induced by dehydration and storage at ambient or cold temperatures have also been identified. Genes changing significantly in expression (analysed using GeneSpring software) as a result of these treatments but not as a result of senescence represent good candidates for the use in diagnostic tests to detect poorly treated flowers. Such tests are important to develop as stored flowers are virtually indistinguishable from freshly harvested flowers and yet their vase life and pigment development (Fig. 3) are severely impaired when placed in the vase at home.
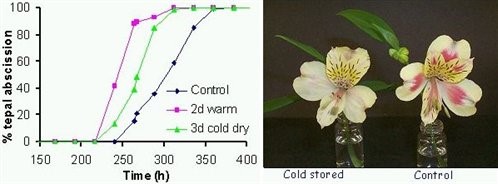
Figure 3. Time course of tepal abscission from control and cold or warm treated flowers (left) and appearance of control and cold treated flowers five days after treatment (right).
During senescence we have investigated the changes in gene expression at four time points during floral development and senescence. Amongst the genes with significantly changed expression patterns that are several which change in expression as a result of cold and warm treatment, others seem to be unique to petal senescence (Fig. 4).
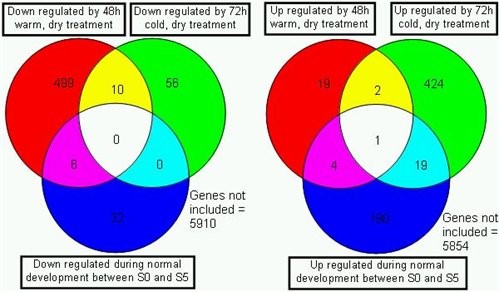
Figure 4. The numbers of genes showing down (left) or up (right) regulation as a result of a 48h warm dry treatment to flowers (upper left circle) or a 72h exposure to cold, dry conditions (upper right circle). In both cases the tissues were briefly refreshed in water after treatment before RNA extraction. In each case the number of genes showing the same expression changes later during development in untreated controls is shown (lower circle).
Floral preservatives clearly improve floral longevity in Alstroemeria as well as other species but little is known about the changes in gene expression that these chemicals induce; using our array we are beginning to unravel the effects that the commonly used preservatives have on gene expression in Alstroemeria (Fig. 5).
Applying the knowledge gained from studies such as these will provide a better basis for the improved formulation of novel preservatives since we will be able to target genes which we know change expression during senescence.
Equally knowing if the up (or down) regulation of particular genes results in longer lived flowers will enable breeding for plants with enhanced (or reduced) expression of these genes and will result in better quality flowers for the consumer. A further goal is to develop diagnostic tools to identify mistreated flowers to ensure that the guarantees offered to consumers in the UK are accurate and realistic. To achieve this we are integrating knowledge of changes in gene expression with techniques such as hyperspectral imaging to provide rapid, non-destructive methods to assess, what in effect are, changes in gene expression.
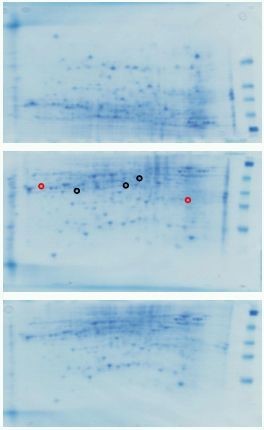
Figure 6. 2D gels of protein extracts from petals just prior to opening (A), 24h after opening (ie fully open) and held in water (B) and after 84h when flowers held in 1mM cycloheximide (ie still appearing fresh and fully open). Black circles indicate spots that differ between A and B and red circles between B and C. Other protein spots may also differ quantitatively or qualitatively.
Research output: Contribution to journal › Article › peer-review
Research output: Contribution to journal › Article › peer-review
Research output: Contribution to journal › Article › peer-review
Research output: Contribution to journal › Meeting abstract › peer-review
Research output: Contribution to journal › Article › peer-review
Nisbet, E., Lowry, D., Basu, A., Stead, T., Lopez Juez, E., Lanoiselle, M. & Fisher, R.
Natural Envt Research Council (NERC)
1/07/17 → 30/06/19
Project: Other
Biotechnology&BioSci Research BBSRC
1/10/14 → 1/10/18
Project: Research